Shutdown & Turnaround Management

The course includes specific checklists, procedures, and strategies designed to enhance your current shutdown planning and execution.
Freeze Drying for Biologics/Pharmaceutics

This practical course will focus on the freeze-drying process from formulation and thermal analysis to cycle development and optimization, packaging and troubleshooting.
Materials and Corrosion in Well Engineering

This interactive course is intended for materials and corrosion specialists, well engineers, production technologists, and other disciplines involved in well design and operations in the energy related sectors.
Effective R&D Project Management in Pharma & Biotech
This course will profile best practice in project management, that is being aspired to by leading organisations.
Pipeline Systems in Energy Transition

The transition to a green energy economy requires a fundamental shift in how critical energy carriers like hydrogen, ammonia, and carbon dioxide are transported.
11th Annual Smart Workspace Design Summit

SWDS is an experience-based event bringing together those who strive for better workspaces. This is where people, place and technology come together to start building the human-centric office of the future, today.
19th Annual Collateral Management and Securities Lending Forum

Collateral Management Forum | 19+ years of financial services events world-wide!
Cell & Gene Therapy Products – CMC & Quality Requirements

With the increasing complexity of new cell & gene therapies, as well as a rapid growth of these products in development, unique challenges have emerged especially in manufacturing and control.
Stability & Shelf Life of Biologics

The purpose of this Stability & Shelf Life of Biologics Training Course is to get comprehensive overview on stability and shelf-life regulatory landscape.
Companion Diagnostics (CDx) – Navigating the Regulatory Challenges

The implementation of the EU In Vitro Diagnostic Device Regulation 2017/746 (IVDR) has introduced new processes for the authorization of performance studies and conformity assessment of companion diagnostic devices (CDx) in Europe.
Pharma & Biotech R&D Portfolio Management in the 21st Century

This two-day training course is designed for portfolio management professionals in Pharma and Biotech who want to enhance their skills and gain insights into the latest tools and techniques for managing complex portfolios.
Inspection and Maintenance of Hydrogen Installations

This course provides a comprehensive examination of the inspection and maintenance of hydrogen installations, covering pipelines, piping systems, machinery, storage systems, and transportation.
Pipeline Systems in Energy Transition

The transition to a green energy economy requires a fundamental shift in how critical energy carriers like hydrogen, ammonia, and carbon dioxide are transported.
Process Analyzers and Sampling Systems

The purpose of this Process Analyzers and Sampling Systems training course is to achieve optimal performance through an understanding of operating principles, possible interference and practical knowledge.
ASME B31.3 Process Piping

This comprehensive four-day course delves into the intricacies of the ASME B31.3 Process Piping Code, equipping participants with a profound understanding of the standards that govern the design, materials, fabrication, installation, inspection, and testing of process piping systems.
Process Safety Techniques with Application on Hydrogen Production Plants
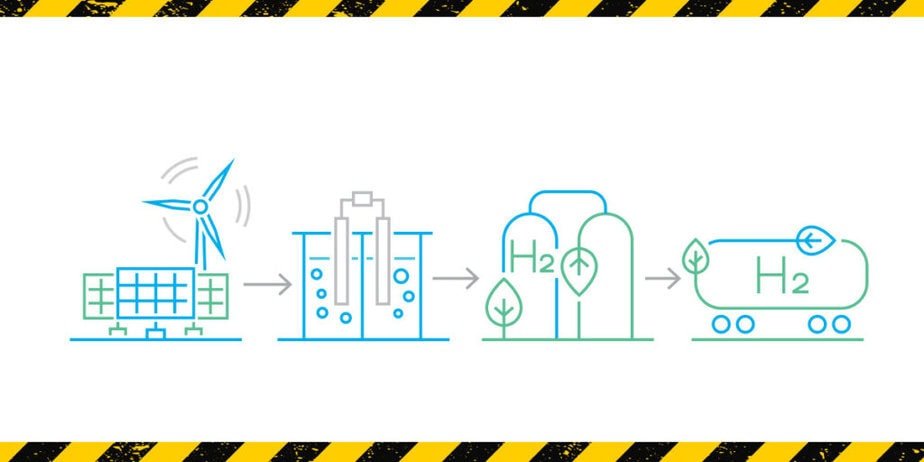
The course presents the essential elements of the activities related to process safety with applications on H2 plants, with particular attention to their contextualization and integration with the management processes during the life cycle of plants.
Process Safety for Hydrogen Carrier Molecules

This course will provide you with the insights into safety challenges and some of the technical challenges that need to be considered and addressed when supply strategies and specific projects are developed.
In-Line Inspection and Inspection of Challenging Pipelines

The course will provide an in-depth introduction into the subject of the in-line inspection of pipelines. We will look at the role the in-line inspection plays in the overall pipeline inspection and pipeline maintenance procedures.
Fitness for Service Assessments

This 5-days advanced course provides guidance for conducting FFS assessments using methodologies as covered in the de-facto industry standard API 579/ASME FFS-1, applicable to the pressurized equipment.
Defect Assessment in Pipelines

This course will present the latest defect assessment methods to pipeline engineers and managers. These methods will range from simple, quick assessment methods, to the more detailed “fitness for purpose” analysis.
